IN HEALTH, AGING, AND DISEASE
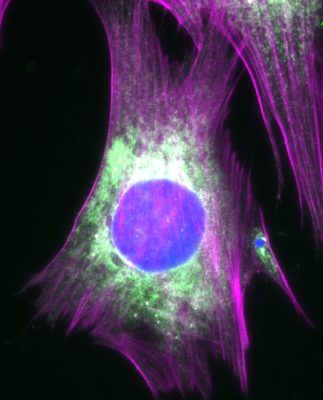
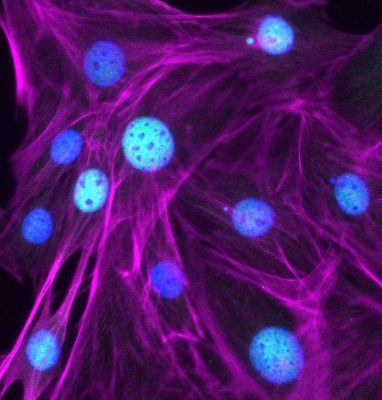
To understand how cells normally function and how they respond to different types of stress, our work focuses on determining: (1) how the cytoskeleton controls the organization, shape, and movement of cells and their internal components; (2) how cytoskeletal remodeling is altered by infectious microbes and genetic mutations; and (3) how cytoskeletal functions affect biological aging. We use a combination of bioinformatic, genetic, biochemical, molecular, cellular, and organismal approaches to study these processes.
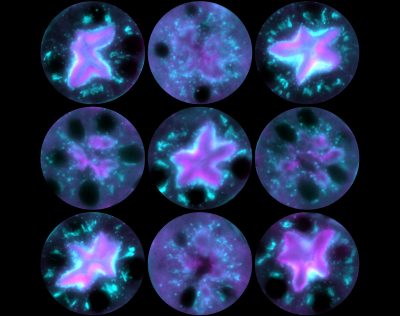
The overarching goal of all the projects in the lab is to understand the mechanisms by which actin nucleation factors govern specific cellular functions. To achieve a comprehensive understanding of the function of the actin cytoskeleton, we need to identify all of the nucleation factors, define their biochemical activities, and determine how their inactivation affects cellular functions. These processes include membrane protrusion, cell motility, biosynthetic transport, endocytic trafficking, autophagy, and apoptosis. In mammalian cells, actin filament networks are nucleated and organized by a macromolecule called the Arp2/3 complex, which works in conjunction with 8 nucleation-promoting factors from the Wiskott-Aldrich Syndrome Protein (WASP) family – WASP, N-WASP, WAVE1, WAVE2, WAVE3, WASH, WHAMM, and JMY – plus several additional atypical nucleation factors including WHIMP, Cortactin, and WISH/DIP1/SPIN90. Other key actin nucleating proteins include 15 members of the Formin family and at least 6 tandem actin monomer binding nucleators from the Spire, Cobl, and Lmod families.
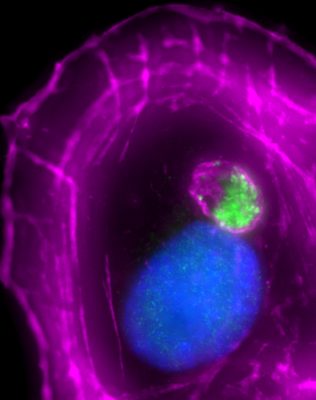
Although determining the basic functions of the actin cytoskeleton is a major component of our research, we are also very interested in understanding how these processes are altered during disease pathogenesis. Due to its role at the foundation of so many cellular functions, the cytoskeleton is often targeted by pathogenic microbes. One such bacterium is enterohemorrhagic Escherichia coli O157 (EHEC), a leading cause of bloody diarrhea and pediatric kidney failure in the US. EHEC is able to modify many membrane remodeling pathways and reorganize cortical actin filaments into protrusive ‘pedestals’ during infection. However, the mechanisms by which most EHEC effectors exploit their host protein targets to elicit these and other phenotypes are not well understood. We are currently investigating the mechanisms that control EHEC virulence factor expression and function.
Science Writing